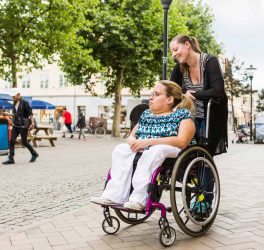Bristol Myers Squibb (BMS) has entered a partnership with US-based non-profit organisation Disability Solutions for the Disability Diversity in Clinical Trials (DDiCT) initiative to ensure equal access to clinical trials, ensuring all patient groups are ‘reflective of the real-world population’.
This new initiative aligns with Bristol Myers Squibb’s broader inclusion and diversity health equity commitments to address health disparities, clinical trial diversity, supplier diversity, employee giving, and workforce representation between 2020 and 2025.
The DDiCT initiative initially aims to make recommendations on how to effectively improve access, engagement, speed of enrollment, and participation of people with disabilities in clinical trials, to ensure all patient groups are reflective of the real-world population and aligned with the epidemiology of the disease studies. This project was initiated by the Bristol Myers Squibb People & Business Resource Group DAWN(Disability Advancement Workplace Network) and will be co-led by DAWN and the Global Drug Development Team.
“Through this work, Bristol Myers Squibb can set the standard and stage for access to life-changing and life-saving medicines for people with disabilities,” said Samit Hirawat, M.D., executive vice president and chief medical officer, Global Drug Development, Bristol Myers Squibb. “The long-term goal of our DDiCT program is to develop and pilot trials that are accessible to the widest variety of patients.”
Current common clinical trial practices exclude up to one-fourth of the U.S. population-based on disability status. According to a study published in the Journal of the American Medical Association, in 338 phase III and IV studies, 12.4% of people with intellectual or developmental disabilities and 1.8% of those with physical disabilities failed to be included due to explicit exclusion criteria. The study also identified that additional barriers for disability diversity within clinical trials are caused by inaccessible trial sites, medical equipment, and ableist biases which deter this community from receiving potentially life-saving treatments.
“People with disabilities are omitted from conversations about diversity and inclusion, despite being the largest underrepresented group in the world and the only underrepresented group anyone can join at any given moment. Therefore, it’s essential that we broaden the scope of medical trials and research,” said Tinamarie Duff, DAWN Global People & Business Resource Group Lead, Bristol Myers Squibb. “The launch of the DDiCT, especially during Disability Pride Month, supports Bristol Myers Squibb’s overall commitment to address every dimension of diversity, which means making the most effective medicine to include people with disabilities at all stages of access/trials.”